Going from more disorder to less disorder decreases entropy. Seto, M., Yoda, Y., Kikuta, S., Zhang, X. Answer: a liquid cooling Explanation: Liquid cooling results in liquid (more disorder) turning to solid (less disorder). 
To rank items as equivalent, overlap them. Rank these systems in order of decreasing entropy. 
Nuclear Condensed Matter Physics Using Synchrotron Radiation (Springer, 2005). At the molecular level, entropy can be described in terms of the possible number of different arrangements of particle positions and. Here, Stot means the change in entropy of a system plus the change in entropy of the. Invar and Elinvar, Nobel Lecture (The Nobel Foundation, 1920). property of entropy is that it never decreases. Now, writing in Nature Physics, Stefan Lohaus and colleagues have reported that a cancellation between the contributions of atomic vibrations and magnetic spins to Invar’s thermal expansion may explain its special behaviour 3. Over the years, various models and theories have been proposed but a full solution to the so-called Invar problem 2 has remained elusive. More recently, large slabs of Invar can be found in many laser optics applications that require any thermal expansion in apparatus to be smaller than optical wavelength scales. Its properties helped to increase the reliability and accuracy of spring watches and enabled the construction of instruments that provide atomic time standards. Explain (a) why the entropy increases and (b) why under most circumstances, a decrease in volume results in an entropy decrease. This leaves us with problem (C) as the correct. At the time of its discovery, this unusual property had a considerable effect on engineering of new instruments for precision metrology, for which its discoverer Charles Édouard Guillaume was awarded the Nobel Prize in Physics in 1920 1. Despite this fact, the entropy of the system increases. Therefore, we can say that entropy (measurement of disorder in a system) always increases and never decreases. 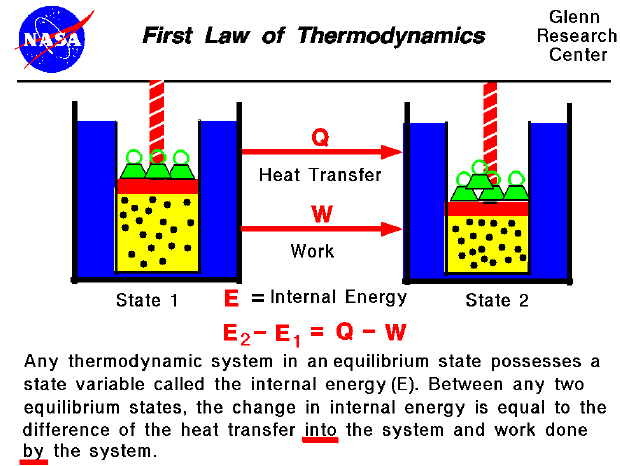
To rank items as equivalent overlap them. Finally, substances with strong hydrogen bonds have lower values of S°, which reflects a more ordered structure.Invar is a magnetic iron–nickel alloy that displays almost no change in length and volume upon change of temperature. The entropy metric is a simple and effective index to design for energy-storage applications. Rank these systems in order of decreasing entropy. For example, compare the S° values for CH 3OH(l) and CH 3CH 2OH(l). Second law: In an isolated system, natural processes are spontaneous when they lead to an increase in disorder, or entropy. First law: Energy is conserved it can be neither created nor destroyed. Similarly, the absolute entropy of a substance tends to increase with increasing molecular complexity because the number of available microstates increases with molecular complexity. Chemical thermodynamics is the portion of thermodynamics that pertains to chemical reactions. Soft crystalline substances and those with larger atoms tend to have higher entropies because of increased molecular motion and disorder. To rank items as equivalent, overlap them. v t e Entropy is a scientific concept, as well as a measurable physical property, that is most commonly associated with a state of disorder, randomness, or uncertainty. In contrast, graphite, the softer, less rigid allotrope of carbon, has a higher S° due to more disorder in the crystal. Transcribed Image Text: Part A Rank these systems in order of decreasing entropy. Among crystalline materials, those with the lowest entropies tend to be rigid crystals composed of small atoms linked by strong, highly directional bonds, such as diamond. \( \newcommand\) also reveals that substances with similar molecular structures tend to have similar S° values. Rank these systems in order of decreasing entropy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
